Drug Excretion
Why do we want to understand excretion pathways?
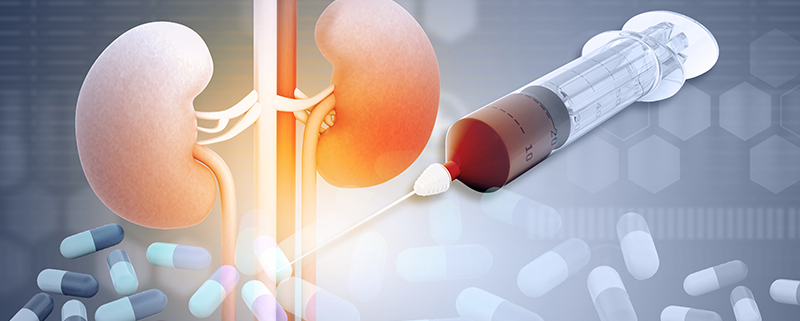
After a drug has entered the body it will be eliminated via a number of routes however, as the FDA renal guidance states, most drugs are cleared by elimination of unchanged drug by the kidney and/or by metabolism in the liver and/or small intestine. If a drug is eliminated primarily through renal or hepatic excretory mechanisms, impaired function can alter the drug’s pharmacokinetics to an extent that the dosage regimen needs to be changed from that used in patients with normal organ function or may even need to be contraindicated.

Mass Balance studies typically administer the investigational drug that has a radiolabeled isotope such as carbon 14 attached. The radiolabel allows total radioactivity of the investigational drug to be tracked, whether it be as the administered parent drug or metabolites. Excreta including urine and feces are monitored for total radioactivity following administration of the radiolabeled investigational drug. This allows us to track the major routes for elimination of the parent drug and metabolites.
Renal Impairment
After a drug has entered the body it will be eliminated via a number of routes however, as the FDA renal guidance states, most drugs are cleared by elimination of unchanged drug by the kidney and/or by metabolism in the liver and/or small intestine. If a drug is eliminated primarily through renal excretory mechanisms, impaired renal function usually alters the drug’s pharmacokinetics to an extent that the dosage regimen needs to be changed from that used in patients with normal renal function.
Renal impairment studies are important when the impact of renal impairment is likely to impact the pharmacokinetics of the drug, for example if greater than 30% of the drug or active metabolite are found in the urine.
Renal impairment can inhibit some pathways of hepatic and gut drug metabolism and transport. Therefore, a PK study in patients with renal impairment should be conducted for most drugs intended for chronic use. Some drugs that are not chronically used can also be evaluated in patients with renal impairment for dose adjustment purposes if there are clinical concerns for use in these patients. There is evidence that even biologics with molecular weights less than 69kDa renal impairment can impact renal clearance of cytokines and cytokine modulators.
Hepatic Impairment
The FDA guidance on hepatic impairment states that, “The liver is involved in the clearance of many drugs through a variety of oxidative and conjugative metabolic pathways and/or through biliary excretion of unchanged drug or metabolites. Alterations of these excretory and metabolic activities by hepatic impairment can lead to drug accumulation or, less often, failure to form an active metabolite.”
From this we can gather that it is important to understand the impact of hepatic impairment on exposures of drugs is needed in order to best understand the impact on efficacy and safety for patients and inform whether doses need to be adjusted in patients with hepatic impairment or whether the treatment cannot be administered in patients with hepatic impairment safely and therefore requires a contraindication listed in the drug label.
The primary purpose of the hepatic impairment guidance is to help understand whether dosage adjustments are required in patients with hepatic impairment based on the impact of hepatic impairment on PK and/or PD of a drug and its active metabolites.
Understanding of elimination pathways and any potential impact of organ impairment on pharmacokinetics, efficacy and safety is critical to a drug development program and also key information to enable safe and effective clinical prescribing in patients. Scientists at PK consultancy have expertise in providing gap analyses and guiding drug development programs from IND to NDA stages. Contact us if you would like to discuss your development program.
References